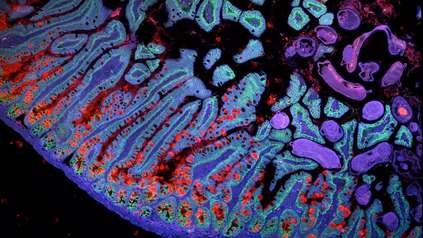
Detailed map of the heart provides new insights into cardiac health and disease
In a new study, published today (12 July) in Nature, researchers have produced the most detailed and comprehensive human Heart Cell Atlas to date, including the specialised tissue of the cardiac conduction system – where the heartbeat originates.
The multi-centre team is led by the Wellcome Sanger Institute and the National Heart and Lung Institute at Imperial College London, and has also presented a new drug-repurposing computational tool called Drug2cell, which can provide insights into the effects of drugs on heart rate.
This study is part of the international Human Cell Atlas* (HCA) initiative, which is mapping every cell type in the human body, to transform our understanding of health and disease, and will form the foundation for a fully integrated HCA Human Heart Cell Atlas.
Charting eight regions of the human heart, the work describes 75 different cell states including the cells of the cardiac conduction system – the group of cells responsible for the heartbeat – not understood at such a detailed level (1) in humans before. The human cardiac conduction system, the heart’s ‘wiring’, sends electrical impulses from the top to the bottom of the heart and coordinates the heartbeat.
By using spatial transcriptomics, which gives a “map” of where cells sit within a tissue, researchers were also able to understand how these cells communicate with each other for the first time. This map acts as a molecular guidebook, showing what healthy cells look like, and providing a crucial reference to understand what goes wrong in disease. The findings will help understand diseases such as those affecting the heart rhythm.
The assembly of a Human Heart Cell Atlas is key given that cardiovascular diseases are the leading cause of death globally. Around 20,000 electronic pacemakers are implanted each year in the UK for these disorders (2). These can be ineffective and are prone to complications and side-effects (3). Understanding the biology of the cells of the conduction system and how they differ from muscle cells paves the way to therapies to boost cardiac health and develop targeted treatments for arrhythmias.
The team also presents a new computational tool called Drug2cell. The tool can predict drug targets as well as drug side effects. It leverages single-cell profiles and the 19 million drug-target interactions in the EMBL-EBI ChEMBL database.
Unexpectedly, this tool identified that pacemaker cells express the target of certain medications, such as GLP1 drugs, which are used for diabetes and weight loss and are known to increase the heart rate as a side-effect, the mechanism of which was unclear. This study suggests that the increase in heart rate might be partly due to a direct action of these drugs on pacemaker cells, a finding the team also showed in an experimental stem cell model of pacemaker cells.
“The cardiac conduction system is critical for the regular and coordinated beating of our hearts, yet the cells which make it up are poorly understood. This study sheds new light by defining the profiles of these cells, as well as the multicellular niches they inhabit. This deeper understanding opens the door to better, targeted anti-arrhythmic therapies in the future.”
Dr James Cranley, joint first author, a cardiologist specialising in heart rhythm disorders and PhD student at the Wellcome Sanger Institute
“The mechanism of activating and suppressing pacemaker cell genes is not clear, especially in humans. This is important for improving cell therapy to facilitate the production of pacemaker cells or to prevent the excessive spontaneous firing of cells. By understanding these cells at an individual genetic level, we can potentially develop new ways to improve heart treatments.”
Dr Kazumasa Kanemaru, joint first author and Postdoctoral Fellow in the Gene Expression Genomics team at the Wellcome Sanger Institute
The study unearthed an unexpected discovery: a close relationship between conduction system cells and glial cells. Glial cells are part of the nervous system and are traditionally found in the brain. They have been explored very little in the heart. This research suggests that glial cells are in physical contact with conduction system cells and may play an important supporting role: communicating with the pacemaker cells, guiding nerve endings to them, and supporting their release of glutamate, a neurotransmitter.
Another key finding of the study is an immune structure on the heart’s outer surface. This contains plasma cells, which release antibodies into the space around the heart to prevent infection from the nearby lungs. The researchers also identified a cellular niche enriching for a hormone (4) that could be interpreted as an early warning sign of heart failure.
“We often don’t fully know what impact a new treatment will have on the heart and its electrical impulses – this can mean a drug is withdrawn or fails to make it to the market. Our team developed the Drug2cell platform to improve how we evaluate new treatments and how they can affect our hearts, and potentially other tissues too. This could provide us with an invaluable tool to identify new drugs which target specific cells, as well as help to predict any potential side-effects early on in drug development.”
Dr Michela Noseda, senior Lecturer in Cardiac Molecular Pathology at the National Heart and Lung Institute, Imperial College London, a Coordinator of the Human Cell Atlas Heart BioNetwork and a lead author
“Using cutting-edge technologies, this research provides further intricate detail about the cells that make up specialised regions of the human heart and how those cells communicate with each other. The new findings on the heart’s electrical conduction system and its regulation are likely to open up new approaches to preventing and treating rhythm disturbances that can impair the heart’s function and may even become life-threatening.
“International collaboration is key to scientific progress. This impactful study and other discoveries from the broader Human Cell Atlas initiative are excellent examples of what can be achieved when the international research community works together across borders. Our combined efforts can ultimately produce better outcomes for patients worldwide.”
Professor Metin Avkiran, Associate Medical Director at the British Heart Foundation, which part-funded the research with the German Centre for Cardiovascular Research (DZHK)
“This Heart Cell Atlas reveals cardiac microanatomy in unprecedented detail, including the cardiac conduction system that enables each heartbeat, and is a valuable reference for studying heart disease and designing potential therapeutics. An important contribution to the global Human Cell Atlas initiative, which is mapping every cell type in the body to understand health and disease, it will form the foundation for a fully integrated HCA Human Heart Cell Atlas. In addition, our suite of computational methods will help identify possibilities for repurposing existing drugs to treat diseases in other tissues.”
Dr Sarah Teichmann, a senior author of the study from the Wellcome Sanger Institute and co-chair of the Human Cell Atlas Organising Committee
More information
Human Cell Atlas
*This study is part of the international Human Cell Atlas (HCA) consortium, which is aiming to map every cell type in the human body as a basis for both understanding human health and for diagnosing, monitoring, and treating disease. An open, scientist-led consortium, the HCA is a collaborative effort of researchers, institutes, and funders worldwide, with more than 2,900 members from 94 countries across the globe. The HCA is likely to impact every aspect of biology and medicine, propelling translational discoveries and applications and ultimately leading to a new era of precision medicine.
More information can be found at https://www.humancellatlas.org/
1. Single cell sequencing is a technology that provides cell-specific genetic information. Multi-omics is a biological analysis approach which uses several techniques to obtain a large quantity of data. This can include data from genomics, proteomics, epigenomics or metagenomics studies, all combined to give a holistic view.
2. NICOR report
3. Mirza, M., Strunets, A., et al. (2013) Mechanisms of Arrhythmias and Conduction Disorders in Older Adults. Clin Geriatr Med. 2012 Nov; 28(4): 555–573. DOI: 10.1016/j.cger.2012.08.005
4. They found that some cells located in the heart produce a hormone called Brain Natriuretic Peptide (BNP) which is measured clinically as a biomarker to detect heart failure. This work shows that even in healthy hearts, a small population of muscle cells produces this substance. By comparing with publicly available data from failing hearts researchers found that these same cells grow in number in failing hearts. This opens the door to new therapeutic opportunities to prevent disease progression by targeting specific cell types.
The researchers used novel sequencing techniques, such as single-cell sequencing to analyse which genes are activated in single cells, and spatial transcriptomics. The research includes data from the hearts of 25 healthy organ donors ranging from 20 to 75 years old. Hearts were donated from 25 organ donors whose hearts were healthy but not suitable for organ transplantation for a number of reasons. We are grateful to the deceased donors and their families for their invaluable gift.
Publication:
Kanemaru, K., Cranley, J., et al. (2023) Spatially resolved multiomics of human cardiac niches. Nature. DOI: 10.1038/s41586-023-06311-1 https://www.nature.com/articles/s41586-023-06311-1
Funding:
This research was supported by joint funding from Wellcome, the British Heart Foundation (BHF) and the German Centre for Cardiovascular Research (DZHK), the Chan Zuckerberg Foundation, the National Institute for Health and Care Research (NIHR) Blood and Transplant Research Unit in Organ Donation and Transplantation, the NIHR Imperial Biomedical Research Centre, the Leducq Foundation, the ERC-advanced grant under the European Union Horizon 2020 Research and Innovation Program, and the Deutsche Forschungsgemeinschaft.